CAR T-cells

CAR T-cells:
High resolution recognition and discrimination with a single reagent
With the increasing importance of CAR T-cell therapy in cancer treatment, ensuring the successful modification and monitoring of these engineered T-cells is crucial.
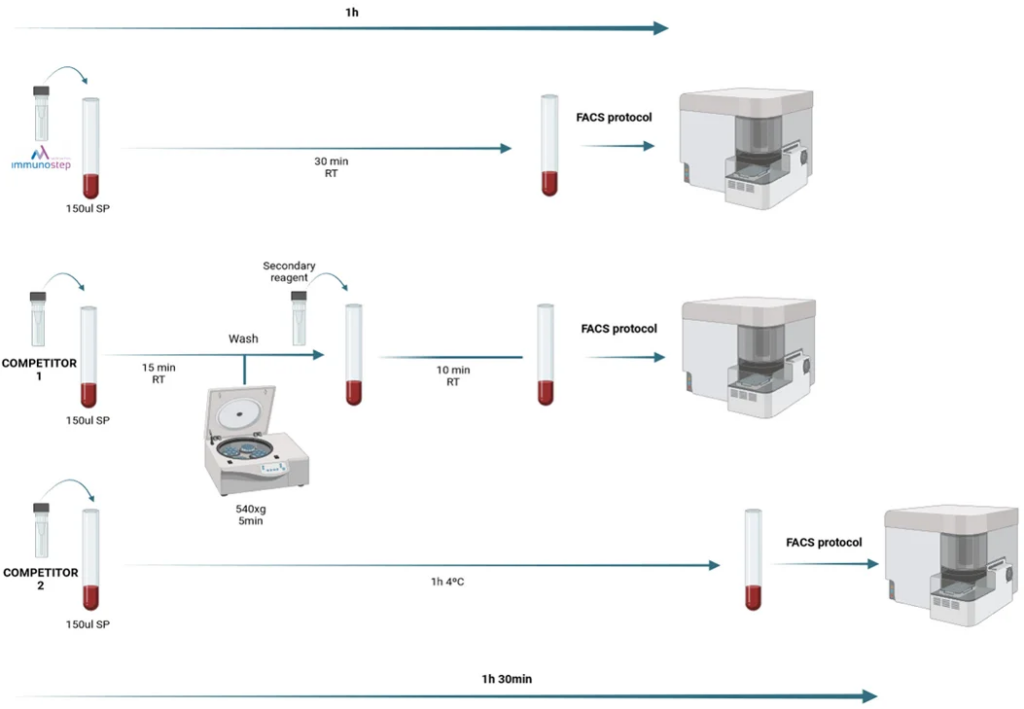
Our Flow Cytometry Reagent simplifies this process, allowing specialists to confirm both key challenges with an easy 1 hour protocol.
Experience the power of precision and efficiency recognising and discriminating CAR T-cells from the rest with high resolution.
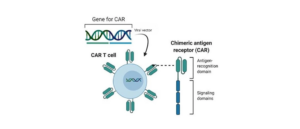
CAR T-cells therapy is based on the extraction of patient T-cells, the introduction of a Chimeric Antigen Receptor (CAR) specific to a protein present on cancer cells (e.g. CD19, BCMA) and the consequently reinfusion of these modified cells, now called CAR T-cells, into the patient to recognise the target cells and eliminate them.
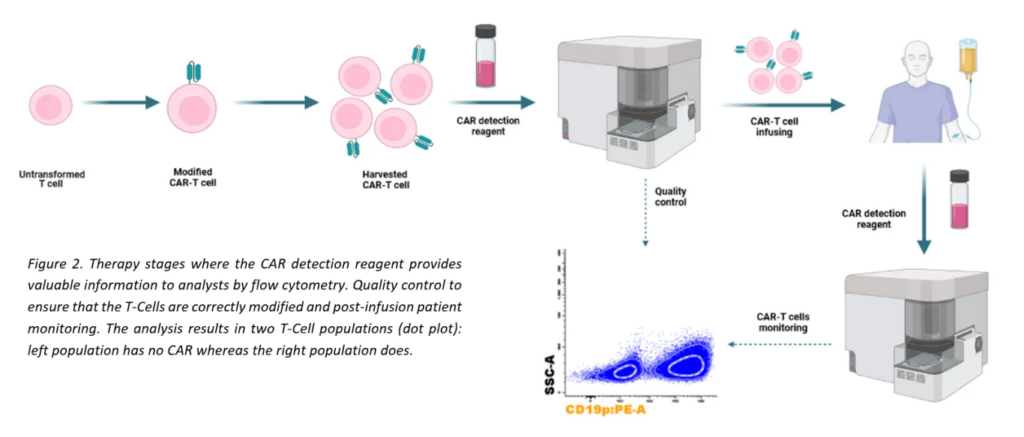
This therapy has gained relevance due to its efficacy, and detecting CAR T-cells tools has become a critical need, both to check that they have been correctly modified, and to monitor them after the reinfusion, since they have a limited life span.
Detection of CAR T-Cells
To meet these needs, we have developed a single reagent for direct and specific recognition of CAR T-Cells with high resolution. This reagent allows the specialists to confirm the successful modification of T-Cells in a quality control prior infusion into the patient and to monitor them after this step.
TECHNOLOGY
Recombinant Fc-Tag CD19 protein (CD19p) and BCMA protein (BCMAp) has been labeled, by a proprietary process, with R-Phycoerythrin fluorochrome (R-PE or PE) or FITC, obtaining a conjugate that is able to be recognized by CAR T-Cells and separates them efficiently without any secondary reagent.
Other manufacturers have similar products based on this fluorochrome-protein complex but have limitations in the separation due to bioconjugation techniques and/or the fluorochrome used.
BrightStepTM: our proprietary labeling technology
This cutting-edge technology is set to redefine CAR-T cell detection. With BrightStep, we’ve improved the art of labeling, ensuring the maintenance of conformation and natural modifications of the protein. This means unparalleled accuracy without compromising the integrity of the sample.
Verified by rigorous flow cytometry testing, Brightstep offers higher performance in specificity and sensitivity. You can trust in its reliability to deliver precise results every time.
Furthermore, with batch-to-batch consistency and reproducibility, you can rely on BrightStep for ensuring reliable outcomes for your research and diagnostics needs.
Experience the future of CAR-T cell detection with BrightStep – where innovation meets accuracy.
| Catalog Number | Name | Size |
| 19-0212-25T | CAR-T cell detection – PE Human protein CD19 | 25 test |
| 19-0212-100T | CAR-T cell detection – PE Human protein CD19 | 100 test |