(CUSTOM) Antikor Geliştirme

Monoclonal And Polyclonal Antibody Development Service
IMMUNOSTEP is able to offer a wide range of high quality customized services for the processes required by its clients. The combination of a number of different technologies that we have developed internally for 15 years means that we can provide our customers with the best solution for their projects.
For the last few decades, the use of both monoclonal and polyclonal antibodies has become widespread in laboratories all over the world. The adaptability of these biological tools todiverse applications means that they are involved in a large number of the processes which are developed in research laboratories, and in the R&D departments of many institutions.
Our 15-year experience in the personalised development of antibodies for our customers has enabled us to take part in the implementation of about 150 of these processes. In turn, they have confronted us with various challenges that have led to the creation of a long list of services related to such developments. Our antibody service portfolio includes conjugation of antibodies, fragmentation, scaling production, development of immunoassays etc.
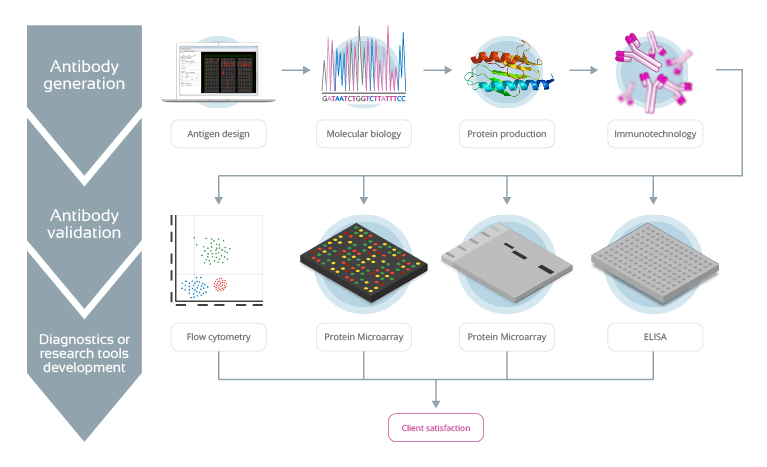
Figure 1: Multidisciplinary programme for the development of customized antibodies
The array of such services allows IMMUNOSTEP to adapt the needs of our customers to our goals with precision.
Polyclonal Antibodies
Our polyclonal antibody development service includes the production of customized polyclonal antibodies in both rabbits and chickens. During the corresponding process, we will work with our clients, and we will adjust our protocols, tested in hundreds of cases, until they have been perfectly adapted to the clients’ needs. This process will not only include the design of the most suitable antigen for a given service, but also peptide synthesis, and animal immunisation protocol. Following on from these steps, the purification and quality control of the antibodies will be implemented by means of an ELISA test.
Rabbit Polyclonal Antibodies
The use of rabbits in polyclonal antibody development has been standard practice for many years. When opting for this kind of process, there are a great many reliable companies for customers to choose from. In the last few years, IMMUNOSTEP has become aware of the fact that what most firms offer seems rather standardised. Moreover, they propose quick solutions, and this does not always fulfil the requirements of the person requesting the service. At IMMUNOSTEP we adapt to your needs within our system.
Depending on the antigen with which the customer wishes to work, we adapt the most suitable one, ranging from straightforward target peptide sequences to the client´s cDNA, utilised as a basis, if available.
Moreover, we have standardised an immunisation protocol that has been thoroughly tested through our experience; this facilitates our knowledge of the behaviour and type of priority Igs which may interest us the most and which allow us “to play around with”the animal´s immune system in order to adapt that response to the technical use the process has been designed for.
Apart from our team’s close relationship with the researcher, another important factor that makes us different is the inclusion in our service of the purification (Protein A/G) of the antibodies from the animals’ serum. This purification allows clients to achieve their goals by working with completely established material right from the start and, most importantly, it provides us with versatility in other services that can help them. For example, specific Ag affinity purification, marked in different fluorochromes/enzymes, subsequent antibody modifications etc. These are additional services that ensure integral and completely personalised assistance.
On occasion, clients will know just what they need, but not which tools are required to do the job. This is where, with the experience we have acquired in hundreds of services, we can help by identifying the necessary tools and delivering them to the customer.

The general time frame for our protocol is as follows:
| Day | Procedure |
| -7 | Arrival of the rabbits |
| 0 | Pre-immune serum collection /1st Immunisation |
| 14 | 1st Booster |
| 24 | Serum collection for titration |
| 35 | 2nd Booster |
| 45 | Serum collection for titration |
| 56 | 3rd Booster |
| 66 | Final bleed |
| Nº | Service | Description |
| 1 | Development of rabbit polyclonal antibody for use ina kit based on “Lateral Flow Immunoassay”. | Development of tools against different pathogens (micro-organisms) for the fabrication of a rapid diagnosis kit. Application of antigens supplied by the client, whole inactivated cells. |
| 2 | Service for the development of rabbit polyclonal antibodies for use against human protein. No Ag supplied by the client. | Study of antigenicity, antigen development based on the client’s needs (WB), peptide synthesis and conjugation with transporting protein. Usual protocol, and testing done by applying the technique requested by the customer. |
| 3 | Service for the development of rabbit polyclonal antibodies against allergens. | Polyclonal antibody development for measuring allergens tested by ELISA. |
| 4 | Service for the development of rabbit polyclonal antibodies against synthetic proteins supplied by clients. | Polyclonal antibody development for the detection of native proteins in WB/IHC for research purposes. |
Chicken Polyclonal Antibodies
The fact that we have been working for many years on finding useful tools for our clients has made us aware of some specific needs that rabbit polyclonal antibodies could not meet: when antigens of interest are highly conserved proteins in mammals, or it coincides that the work of interest is in species which are related phylogenetically.
The use of different animal species is common in polyclonal development, but the structural characteristics of Immunoglobulins Y (IgY) have led to an increase in the utilisation of chickens in the last few years.
IgYs, not present in mammals, have a structure of the Fc region of the Ig which is completely different from that of the IgGs found in mammals. This constitutes a number of advantages when working with them:
– It reduces the background in diverse techniques (WB, IHC etc.) since IgYs do not bind to different mammalian antibodies such as Rheumatoid factor, HAMA etc.
– The mammalian complement system is not activated.
– The IgY does not bind to mammalian Fc receptors.
– The IgY does not attach to Ig binding sites of different natural proteins, such as Protein A (Staphylococcus aureus), Protein G (Streptococcus sp.) or Protein L (Peptostreptococcus magnus).
Moreover, the medium from which IgYs are extracted is not blood, but eggs; thus the animals will not bleed to death, but remain alive.

| Nº | Service | Description |
| 1 | Service for the development of chicken polyclonal antibodies against allergens. | Development of polyclonal antibodies for utilisation in measuring allergens by means of ELISA. |
| 2 | Peptide design and service for the development of chicken polyclonal antibodies for blocking the functional region of Presenilin in humans. | Functional and antigenicity analysis of protein involved in the study of neurodegenerative diseases. Design of blocking tools for in vitrostudies. |
| 3 | Peptide design based on point mutation CKit mut and chicken polyclonal development. | Design of (mutated) sequence and polyclonal antibody development for the detection of highly conserved proteins, C-Kit. |
| 4 | Chicken polyclonal development service against synthetic proteins supplied by clients. | Polyclonal antibody development for the detection of native proteins in WB/IHC for research purposes. |
Polyclonal Antibodies Services
| Study and Analysis for Protein Antigenicity Sequence selection | |||||||
| Immunization | Synthesized | 3 Peptides | 2 Peptides | 1 Peptides | 3 Peptides | 2 Peptides | 1 Protein |
| Antigens | (Customer Supply) | ||||||
| Immunized | 6 (2 for Peptide) | 4 (2 for Peptide) | 2 | 2 | 2 | 2 | |
| Rabbits | |||||||
| Deliverables | Preimmune Serum | 5-10 ml | |||||
| Intermediate Bleedings | 2 Intermediate Bleedings along the Immunitation Protocol – 5-10ml | ||||||
| Quality Control | Complete information Mass Spectrometry and HPLC – Peptide Report | ||||||
| Sample Antigen | 10-15 mg each Antigen (Peptide) | Customer´s Protein | |||||
| PoAb purified Protein G/A (20-30 mg/antigen) | 6 (Total 10-15 mg each Animal) | 4 (Total 10-15 mg each Animal) | 2 (Total 10-15 mg each Animal) | 2 (Total 10-15 mg each Animal) | |||
| Application | Antigen ELISA Titer 1:50000 | WB – Antigen | |||||
| Positive Antigen | |||||||
| ELISA Titer 1:50000 | |||||||
| Guaranteed | Final Report – Calendars, Operations, and any deviation QC. | ||||||
| Amount Antibody | Total 60-90mg Ab | Total 40-60mg Ab | Total 20-30 mg Ab | Total 20-30 mg Ab | |||
| (6 different PoAb) | (4 different PoAb) | (2 different PoAb) | (2 different PoAb) | ||||
| Timeline | 60 days | ||||||
Phospho-Specific Antibodies
At IMMUNOSTEP, we are taking more steps towards adjusting our services to our clients’ needs. Work with new proteins and advances in functionality discoveries sometimes make it necessary to differentiate between the activation/functionality of markers. Post-translational modifications in proteins, especially the phosphorylation of proteins, are involved in most cell processes and this makes their study essential in the development of new strategies in regard to numerous human diseases. Our phospho-specific antibody purification system can detect specific phosphorylations in proteins from complex high sensitivity and high specificity cell mixtures. The high sensitivity and high specificity of IMMUNOSTEP’s phospho-specific antibodies is achieved through thoroughly tested and optimised screening as well as intensive work.

| Nº | Service | Description |
| 1 | Service for the development of chicken phospho-specific polyclonal antibodies against peptide p38α. | Synthesis of phosphorylated peptides (Tyr323) and non-phosphorylated peptides, and production of chicken polyclonal antibodies. Phospho-specific purification. Development for autoimmune diseases. |
| 2 | Service for the development of phospho-specific monoclonal antibodies against peptide Y41. | Production of monoclonal antibodies against peptide Y41. Differential screening process for phosphorylation and non-phosphorylation. |
| 3 | Service for the development of rabbit phospho-specific polyclonal antibodies against peptide TAU. | Synthesis of phosphorylated and non- phosphorylated peptides, and development of rabbit polyclonal antibodies.Phospho-specific purification. |
Monoclonal Antibodies
For the last fifteen years, IMMUNOSTEP has generated dozens of projects in which the development of mouse monoclonal antibodies has been the key to our clients reaching their goals. We can guarantee high affinity and high specificity antibodies that will help our customers to succeed in achieving the aims of their research.
By adapting our standardised production system by means of multiple custom-designed screening processes, we can obtain the tool that the client had in mind.
From a large variety of antigens, which can be developed internally, or from the clients’ previous developments, we will find the solution they require. Using proteins as antigens, we will obtain monoclonal antibodies against the conformational epitopes of the target protein. This will maximise the antibody’s chances of binding to the native protein in a greater range of applications. Thus, from the sequence of this target protein only, we can carry out the whole process internally, and control and design the complete chain of development together with the client.
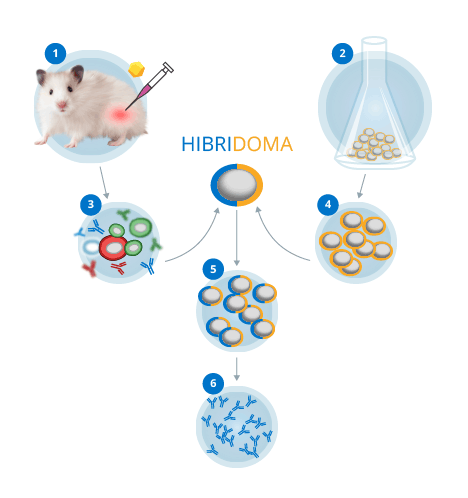
PHASE 1: Study and Immunisation Protocol
It is the key phase in the entire process. Here the client’s needs are established, and subsequently the whole process is designed so as to obtain both the ideal antigen for the development and the control points. The immunisation period will vary from 30 to 90 days as needed.
 Standard protocol or customized design.
Standard protocol or customized design.
 Number of animals: 5 Balb/c mice.
Number of animals: 5 Balb/c mice.
 Intermediate bleeding for immunisation control.
Intermediate bleeding for immunisation control.
PHASE 2: Cell Fusion, Selection and First Screening
An SP2 murine cell line is used to obtain hundreds of clones through thebinding of the splenocytes of immunised animals. These clones will provide a starting point for work on the localisation of the antibody required by the client.
– Conventional ELISA and/or WB for screening
– Validation with the immunogen
– Capture ELISA
– Competitive ELISA
– FACS screening
– Other additional types of screening are available
PHASE 3: Cloning and Second Screening
Once these antibodies/clones have been localised, we will begin to work on them in order to adjust the antibody, now a researcher’s tool.
– Delivery of supernatants to the customer and consensual selection of the candidate clones. Customers make their choice according to their own tests.
PHASE 4: Stabilisation and Cryopreservation. Characterisation of the selected clones.
Tests such as Ig Isotyping, Mycoplasma tests etc.
Monoclonal Antibodies Services
| Protein | Protein | Peptide | Peptide | Peptide Service | |
| Service | Service | Premium 1 | Premium 2 | ||
| Premium | Service | Service | |||
| Guaranteed | WB – Antigen Positive | Antigen ELISA Titer 1:50000 | |||
| Antigen ELISA Titer 1:50000 | Study and Analysis for Protein Antigenicity | ||||
| Sequence selection | |||||
| Immunization | NCBI Acession Number Protein (or cDNA for Protein). | NCBI Acession Number Protein | |||
| Protein synthesized by IMMUNOSTEP | |||||
| Customer Supply | 3 Peptides | 2 Peptides | 1 Peptides | ||
| 3-5 Balb/c Mices | |||||
| Costumer Screening | Shipment to customer´s laboratory | Shipment to customer´s laboratory 4-6 ELISA | |||
| 5-10 ELISA Positive-Antigen | Positive-Antigen supernatants (5-10 ml each clone) | ||||
| supernatants (5-10 ml each clone) | |||||
| Deliverables | Up to 5 Clones (Hybridoma Cells), 2mg MoAb each Clone and 200μg Protein | Up to 5 Clones (Hybridoma Cells), 2mg MoAb each Clone | Up to 10 Clones (Hybridoma Cells), 2mg MoAb each Clone and 5mg each peptide | Up to 8 Clones (Hybridoma Cells), 2mg MoAb each Clone and 5mg each peptide | Up to 6 Clones (Hybridoma Cells), 2mg MoAb each Clone and 5mg each peptide |
| Final Report – Isotyping clones Free Mycoplasma Certifyed | |||||
| Timeline | 9-10 Months | 6-8 Months | 7-9 Months | ||
How can we help you?
For many years our in-house, and in some cases external, antibody production has enabled us to develop an infinite number of solutions for adapting this protocol to each different application. Constant improvements in antigen design, as well as in immunisation processes, and new means of screening in both cases, means that any requirements in new production have been covered.
| Nº | Service | Description |
| 1 | Service for the development of specific antibodies against proteins involved in mitochondrial energy metabolism | |
| Development of antibody pairs to be utilized in the creation of protein arrays. Design and production of antibody pairs within the framework of a project aimed at finding cancer-related mitochondrial energy pathways | ||
| 2 | Service for the design and development of specific antibodies against new protein tags | Development of new monoclonal antibodies against new tags. After designing a new family of tags to be used as advanced tools in protein functional analysis, the battery of specific antibodies was developed for research purposes |
| 3 | Service for the development of specific antibodies against allergens | Design and development of antibody pairs against common environmental allergens for the subsequent production of detection kits for such allergens |
Rico-Leo EM. Et al. Dioxin Receptor Expression Inhibits Basal and Transforming Growth Factor β-induced Epithelial-to-mesenchymal Transition. J Biol Chem. 2013 Mar 15; 288(11): 7841–7856
Sanchez-Blazquez P et al. Mu-Opioid Receptors Transiently Activate the Akt-nNOS Pathway to Produce Sustained Potentiation of PKC-Mediated NMDAR-CaMKII Signaling. PLoS One. 2010; 5(6): e11278.
Muñiz LM et al. Atypical response regulators expressed in the maize endosperm transfer cells link canonical two component systems and seed biology. BMC Plant Biol. 2010; 10: 84.
Reyes D. et al. Immunolocalization of FsPK1 correlates this abscisic acid-induced protein kinase with germination arrest in Fagus sylvatica L. seeds. Journal of Experimental Botany, Vol. 57, No. 4, pp. 923–929, 2006
Morales-Hernandez A et al. Alu retrotransposons promote differentiation of human carcinoma cells through the aryl hydrocarbon receptor.2016 Nucl. Acids Res.
Lara Manyes, et al. Transcriptional profiling reveals functional links between RasGrf1 and Pttg1 in pancreatic beta cells. BMC Genomics. 2014; 15: 1019
“Ricardo Jara-Acevedo, et al. Evaluation of chemically modified carrier proteins for developing monoclonal antibodies against a clinically relevant mutation of cKIT. J Immunol Methods 384 (2012) 171–176″
Maya V Georgieva, et al. Inntags: small self-structured epitopes for innocuous protein tagging. Nature Methods, 2015 doi:10.1038/nmeth.3556





